Clinical Trials Dashboard
About Project
Title
Clinical Trials Dashboard
Medosome Biotec/Freelance
RoleI was the Product Designer throughout the design cycle, from planning and requirements gathering to strategy, wireframing, and design.
SkillsBest in Practice Research, Discovery Research, Interviews, Persona, Survey, Information Architecture, Sitemaps, User Flows, Wireframes, User Testing, UI Design
Problem Statement
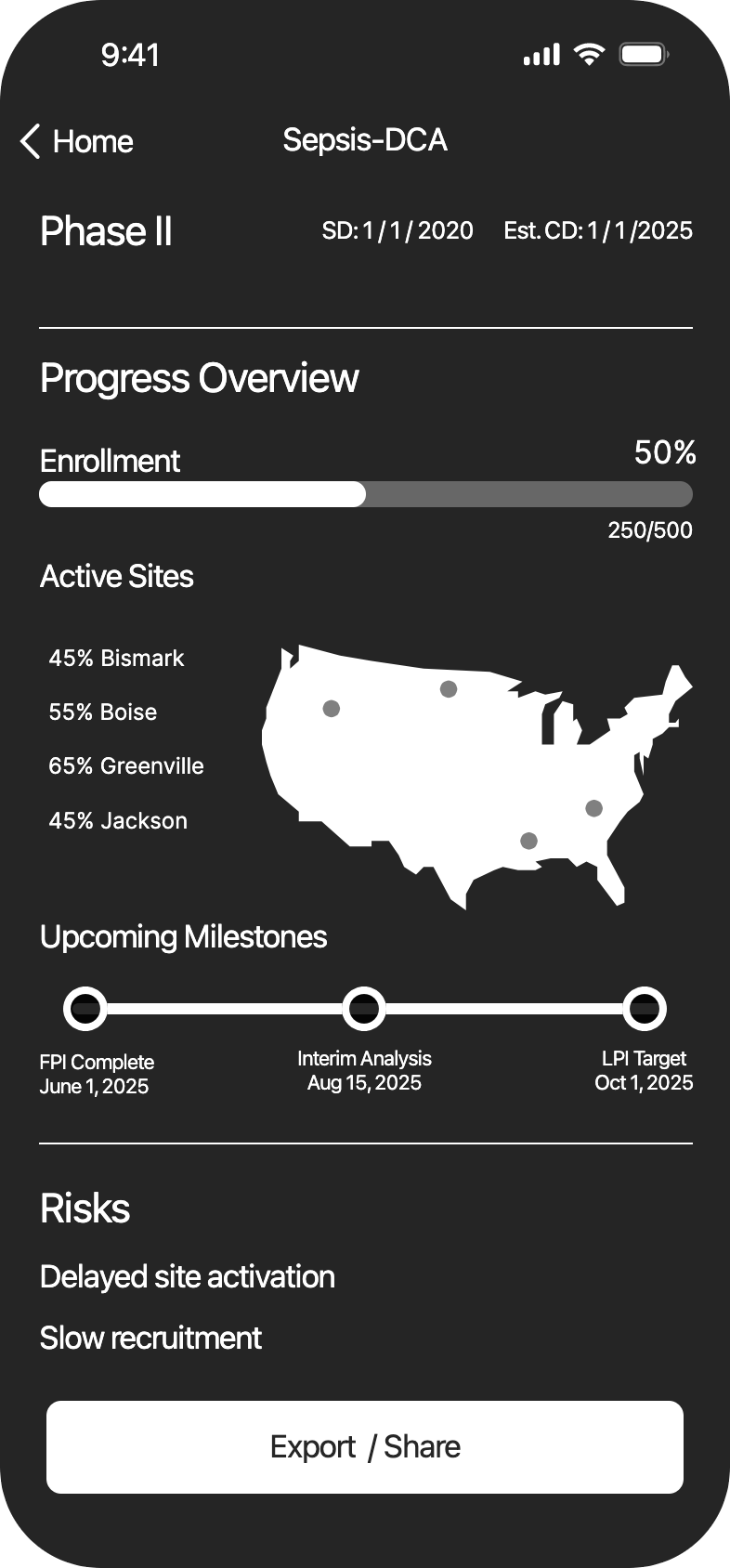
The CEO has multiple clinical trials to track. Money has been lost when leadership was unaware of problems, and was unable to act in time.
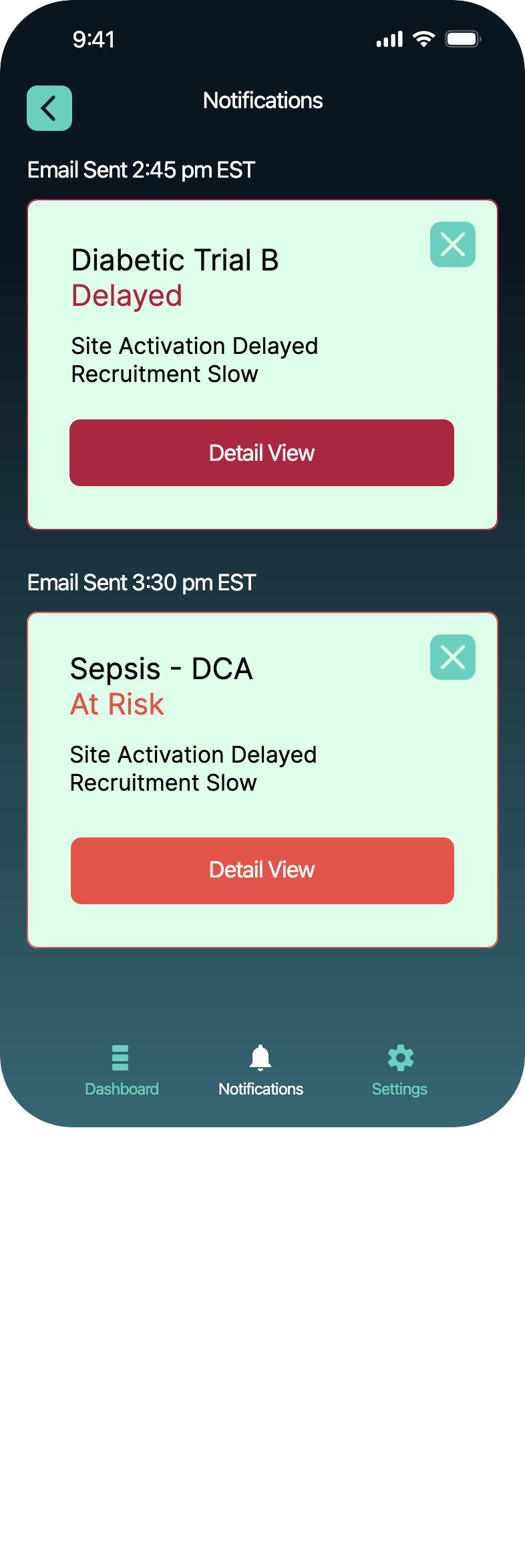
ResearchStakeholder interviews showed that important data is lost in multiple Excel sheets.
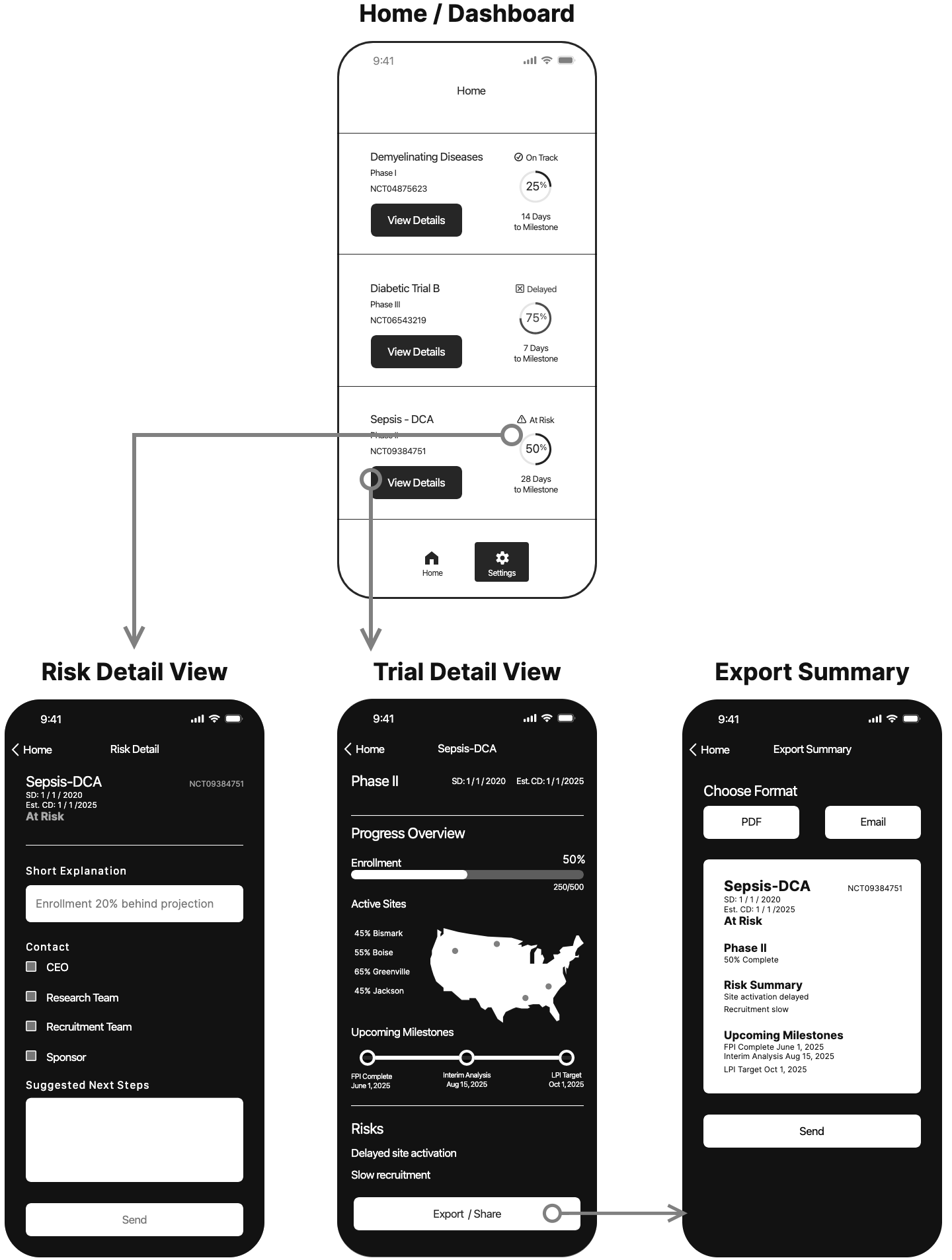
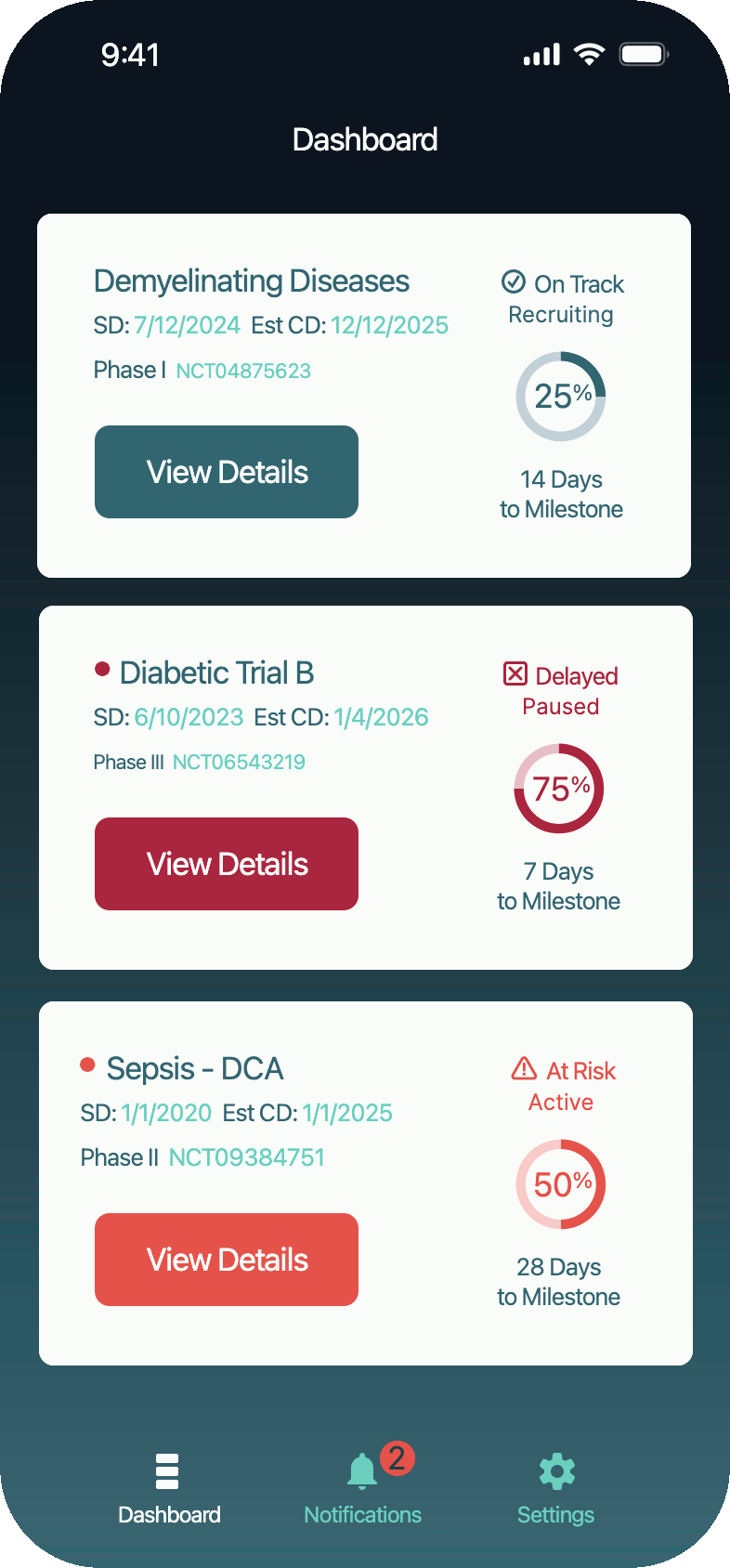
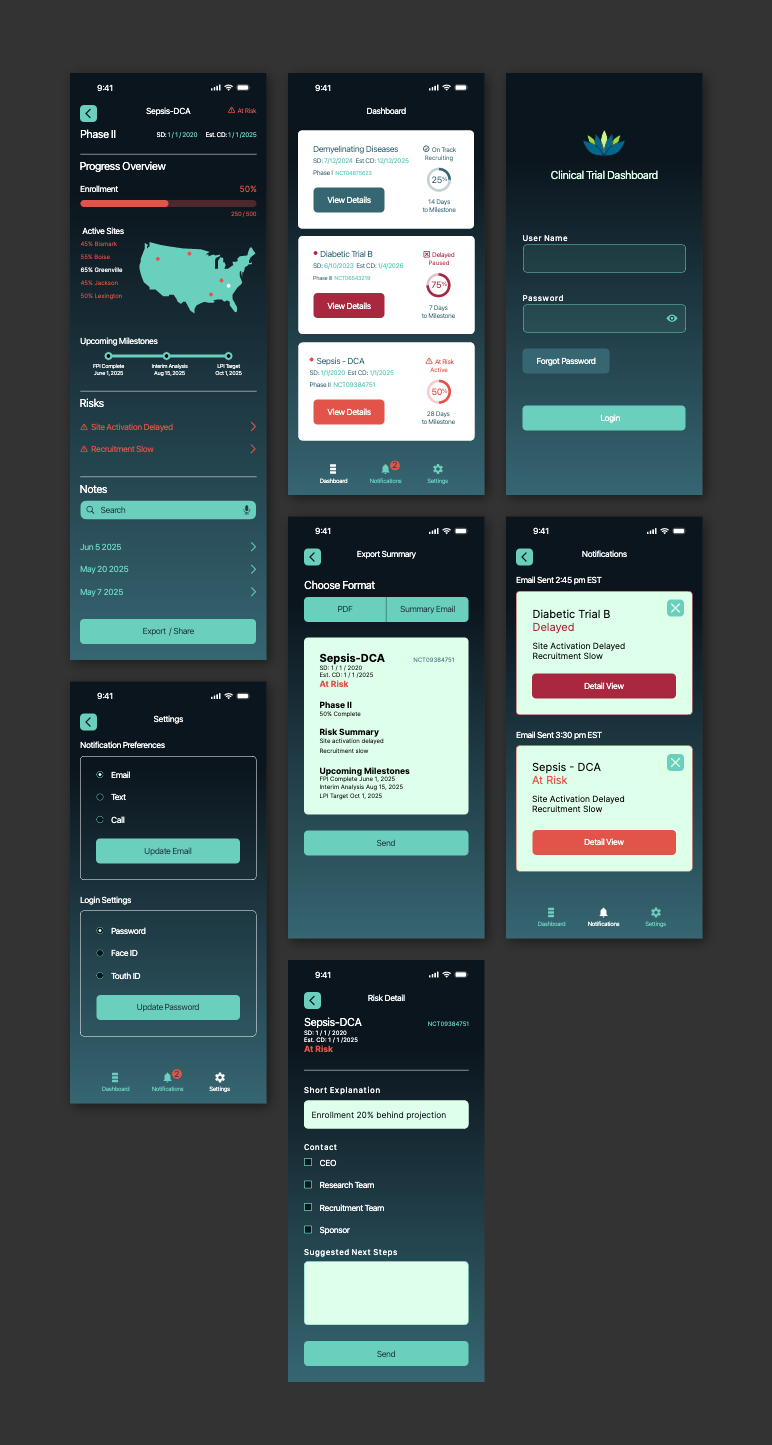
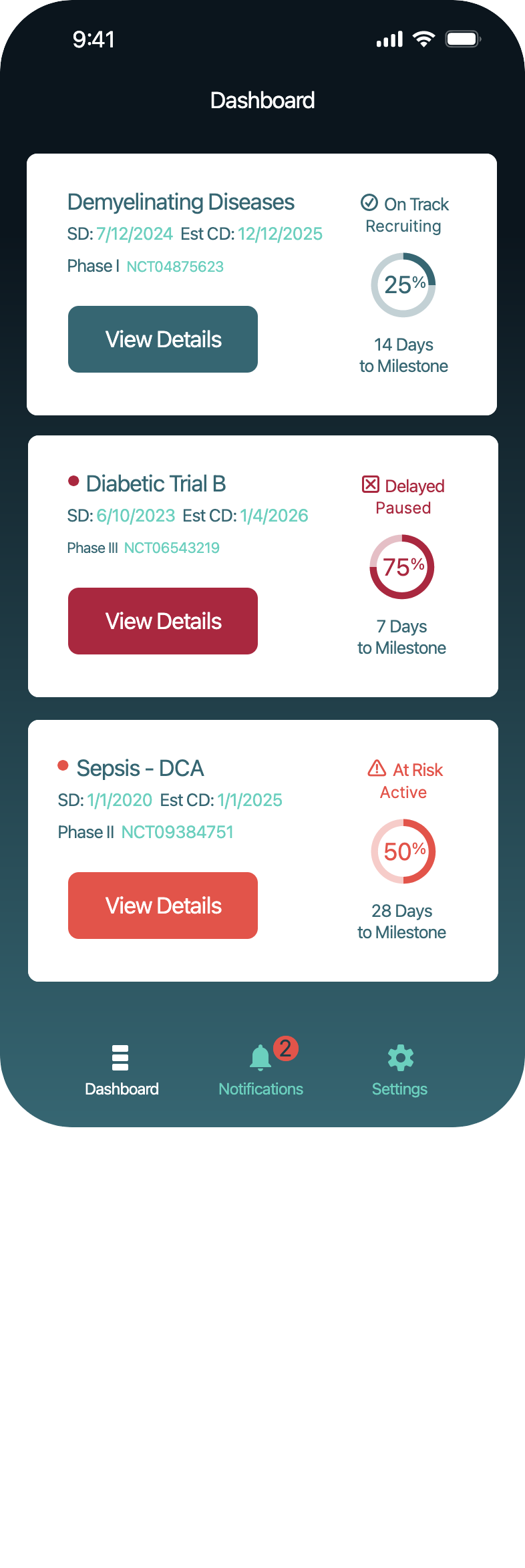
SolutionKey data was made scannable in a dashboard, notifications were sent when pivotal decisions had to be made.
Impact0% Financial Loss: Costly losses due to lost and late information have ended.
Faster, Confident Decision Making: The CEO can intervene early with investor updates, partnerships, or trial pivots. Analysis time reduced 15%.
Increased Trial Oversight Without Added Meetings: Meetings shortened 30% due to clear and shared understanding.
Challenge
The CEO oversees multiple complex clinical trials, but critical data is scattered across numerous spreadsheets.
Core Problem
Data fragmentation causes delayed awareness of issues, leading to missed opportunities for intervention.
Business Need
With limited time and high stakes, the CEO needs a streamlined solution that delivers clear insights, enabling quick, and confident decisions.
Goal
Design a focused application that enables a biotech CEO to monitor real-time progress across clinical trials, quickly, clearly, and in the palm of their hand.






 Above
Above